Page 21 of 23

Chem-Cross Symbol
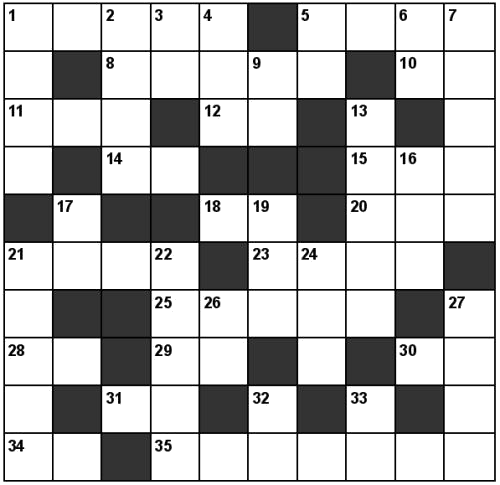
|
Accross |
| 1. Methyl Alcohol. 5. Caustic Soda. 8. The source of Bromine in sea water. 10. Atomic weight of sodium. 11. Formed when aluminium reacts with nitrogen. 12. Element associated with Madame Curie. 14. Allotrope of oxygen. 15. The weight of 8 atoms of calcium. 18. Hydroxyl group. 20. Phosphine. 21. The substance from which hydrogen peroxide is usually prepared in the lab. 23. 2CO+O2= 25. anhydrite. 28. Compounds containing this element give a red flame on a platinum wire. 29. A very light metal. 30. NH4NO2= _ +2H2O 31. Tungsten carbide. 34. At. weight of calcium. 35. Two molecules of the compound which when heated gives nitrous oxide (laughing gas). |
|
Down |
| 1. Aluminium Bronze. 2. 3 molecules of manganous oxide. 3. -xy-en - a gas. 4. The gas prepared by the action of bromine on moist red phosphorus. 5. 3Mg+_ = Mg3N2 6. 2KNO3 = 2KNO2+_ 7. Phosphorous acid. 9. See 12 across. 13. Phosphoric acid. 16. _ +O2 = 2H2O 17. Lanthanum. 19. Sulphuretted hydrogen. 21. The white precipitate formed when barium chloride is added to a solution of a sulphate. 22. Two molecules of the substance on which water reacts to give acetylene. 24. CaCO3 = CaO + _ 26. The metal obtained from the electrolysis of alumina. 27. Nitrogen trioxide. 32. Carbohydrates contain the elements _ , _ and oxygen. 33. Cyanide group. |
